Research & Development
Anti-CCL24 Therapeutic Platform
Introduction
Chemomab’s R&D expertise, infrastructure and capabilities have supported the discovery and development of novel therapies targeting the soluble cytokine CCL24. Our R&D team has investigated the underlying biology of CCL24 and the fibro-inflammatory cycle using a broad range of in-vitro and in-vivo models that have provided the foundation for the development of potentially transformative therapeutics.
Chemomab leverages collaborations with leading investigators from prestigious research centers around the world to obtain further evidence of CCL24’s unique role in fibrosis and inflammation and demonstrate the anti-fibrotic and anti-inflammatory effects of nebokitug, Chemomab’s first-in-class CCL24 neutralizing antibody.
The Roles of CCL24 and Nebokitug
About CCL24
Over the last 15 years, our scientific team has discovered the role of the soluble protein CCL24 as a key regulator in fibro-inflammatory processes, specifically, its pivotal pro-fibrotic and pro-inflammatory effects in fibrotic diseases.
Chemomab researchers have documented CCL24 up-regulation and its correlation with disease severity in fibrotic disease tissues, including liver, skin and lung fibrosis-related pathologies. Based on these discoveries, Chemomab established a comprehensive preclinical package to support the therapeutic effects of blockading CCL24.
We have demonstrated that CCL24 works through a dual biological pathway directly activating fibroblasts while recruiting immune cells and thereby maintaining the inflammatory environment that supports fibrogenesis. Chemomab has shown that inhibiting these pathways by blocking CCL24 prevents and potentially reverses fibrotic tissue growth in the liver, skin and lung.
Based on patient data and a variety of relevant disease models, Chemomab developed nebokitug to target and neutralize CCL24, ameliorating its overexpression and blocking its harmful effects.
About Nebokitug Therapeutic Platform
Nebokitug is a novel therapy that addresses the high unmet need for effective treatments for fibrotic diseases. In preclinical studies, treatment with nebokitug demonstrated strong anti-fibrotic effects, reduced inflammatory injury and significantly improved organ damage. Numerous in-vivo, in-vitro and ex-vivo studies showed that nebokitug demonstrates potent anti-CCL24 effects in disease models of primary sclerosing cholangitis (PSC), systemic sclerosis (SSc), metabolic dysfunction-associated steatohepatitis (MASH) and idiopathic pulmonary fibrosis (IPF). In 5 clinical trials to date, nebokitug appeared safe and well tolerated in healthy subjects and in patients with metabolic dysfunction-associated fatty liver disease (MALFD), MASH, acute lung injury and PSC.
Based on its broad, robust biological effects, favorable tolerability profile, unique mechanism of action and extensive preclinical and clinical data, Chemomab believes nebokitug may have the potential to interrupt the deleterious pathological processes that drive fibrosis, with the potential to become a disease-modifying anti-fibrotic therapeutic agent.
Nebokitug Reduces Both Inflammation and Fibrosis by Neutralizing CCL24
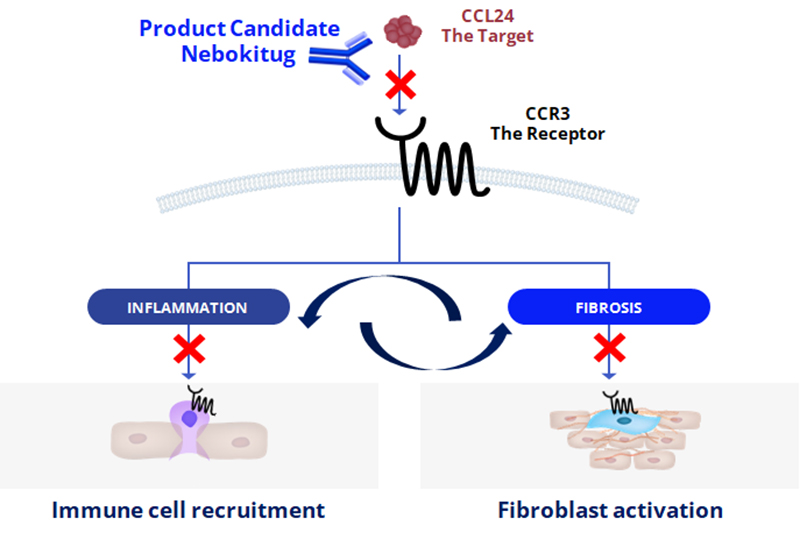
CCL24 Dual Role in Promoting Fibrosis & Inflammation
Neutralizing CCL24 Has Advantages Over Blocking the Receptor
Nebokitug Phase 2 SPRING Trial in Primary Sclerosing Cholangitis
Chemomab’s successful SPRING trial represented the first major clinical proof-of-concept of the anti-fibrotic, anti-inflammatory and anti-cholestatic activity of our CCL24-neutralizing antibody nebokitug. The Phase 2 study (NCT04595825) was a double-blind, placebo-controlled, multiple dose study assessing the safety and tolerability of nebokitug administered to PSC patients with established large duct disease. The trial treated 76 patients in the U.S., Europe and Israel. Patients received either 10 mg/kg or 20 mg/kg of nebokitug or placebo via an intravenous infusion every three weeks over the 15-week treatment period. The study analysis included assessments of all study completers, as well as a prespecified subgroup analysis of moderate/advanced patients with a higher risk of more rapidly progressing disease, defined as patients with a transient elastography (VCTE) measure at baseline of greater than 8.7 kPa, a well-accepted indicator of more progressive PSC disease. Approximately half of the SPRING study patients were classified as having moderate/advanced disease, which is similar to the overall PSC patient population. In addition to safety, the trial measured a wide range of secondary outcomes including serum biomarkers and physiological parameters. These include well-validated liver biomarkers including liver stiffness, ELF and PRO-C3, as well as pruritus and liver function tests.
The SPRING trial included an open label extension (OLE) that was available to those study participants who completed the double-blind portion of the study once the OLE had been established. Of those patients eligible to roll over to the OLE, 93% elected to participate. Patients in the 10 mg/kg and 20 mg/kg dose arms continued on those doses, while placebo patients received either a 10 mg/kg or 20 mg/kg dose based on their initial double-blind assignment. Nebokitug was administered by infusion once every three weeks for an additional 33 weeks, with patients receiving a total of up to 48 weeks of treatment. In general, the OLE analysis assessed most of the same primary, secondary and exploratory endpoints that were measured in the double-blind portion of the trial.
Summary of SPRING Trial 15-Week Results
In the 15-week double-blind portion of the SPRING trial, treatment with nebokitug achieved its primary endpoint of safety and tolerability and demonstrated clinically-relevant anti-fibrotic, anti-inflammatory and anti-cholestatic effects across a broad range of disease-related secondary endpoints, including a statistically significant improvement in liver stiffness, a key PSC disease marker.
Nebokitug-treated patients with moderate/advanced disease showed improvements on multiple disease-related secondary endpoints, including assessments of changes from baseline relative to placebo at Week 15 in liver stiffness (transient elastography); in liver fibrosis biomarkers, including the Enhanced Liver Fibrosis (ELF) score and PRO-C3 levels; in total bilirubin and liver function tests; in pruritis (itch) and in markers of inflammation. Dose-dependent responses were observed for many of the disease-related biomarkers. A consistent pattern of greater improvement on the secondary endpoints was observed in the study arm receiving 20 mg/kg of nebokitug and in the prespecified subgroup of PSC patients with moderate/advanced disease.
Summary of SPRING Trial 48-Week Results
Results from the open label extension (OLE) portion of the SPRING trial confirmed that treatment of PSC patients with nebokitug for up to 48 weeks was safe and well-tolerated and resulted in continued improvements in key liver biomarkers such as the ELF score, the fibrosis-related components of ELF and PRO-C3. Liver stiffness scores were substantially lower in the nebokitug-treated patients with moderate/advanced disease compared to historical controls. Cholestasis-related markers stabilized over 48 weeks of treatment and total serum bile acids were reduced. OLE patients with moderate/advanced disease treated with nebokitug for up to 48 weeks showed a significantly lower number of clinical events (4.8%) compared to historical controls (25.8%). This is especially noteworthy since the primary endpoint of the proposed nebokitug PSC Phase 3 trial design is a composite of PSC-associated clinical events.

